- Home
- Resources
- Combined Assays
- PhenoSense® GT
Monogram Biosciences

PhenoSense® GT
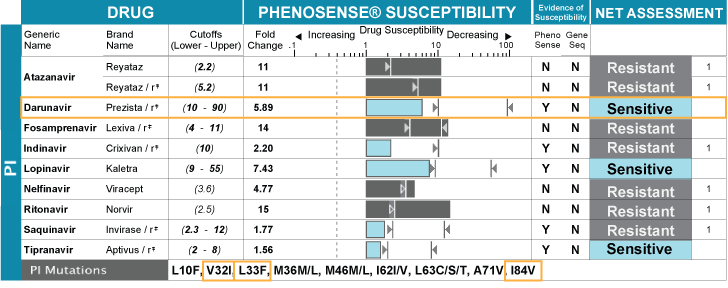
PhenoSense® GT is a combination resistance test available today that provides genotypic information with true phenotypic observations together on the same report.
Only PhenoSense GT provides a complete picture of resistance and brings clarity to health care providers’ treatment choices.
Both phenotypic and genotypic results come from the same blood sample (viral load ≥500 c/ml) and are reported in an easy-to-read format complete with updated upper and lower clinical cutoffs that define the phenotypic susceptibility recommendations. PhenoSense GT also includes HIV-1 subtype and a measure of replication capacity (RC) that provides information about the capability of the patient’s virus to replicate in the absence of drugs. This comprehensive test gives health care providers advanced, practical insight into viral susceptibility, which is why it is the preferred resistance test to optimize background therapy in clinical trials.

Choosing PhenoSense GT after two regimen failures allows health care providers to retain options in each patient’s drug regimen strategy that may preserve drug choices for a later time. While sophisticated in its technology, PhenoSense GT remains user friendly. The report form includes updated upper and lower clinical cutoffs and a summary page.
PhenoSense GT Technology Features
- The largest proprietary database, updated regularly
- More than 100,000 matched phenotypic and genotypic pairs
- Accurate and direct measure of resistance for reliable results
- Identification of optimal drug choices
- Resolves discordance between actual phenotype and genotype with a net assessment
- Appropriate for the evaluation of patient specimens with HIV-1 viral loads ≥ 500 copies/mL